Application of the nourseothricin acetyltransferase gene (nat1) as dominant marker for the transformation of
filamentous fungi
Ulrich Kück and Birgit Hoff
Lehrstuhl für Allgemeine und Molekulare Botanik, Ruhr-University Bochum, 44780 Bochum, Germany
Fungal Genetics Newsletter 53:9-11
Here, we report the construction of two transformation vectors, pD-NAT1 and pG-NAT1, carrying the nat1 gene
encoding the nourseothricin acetyltransferase. The nat1 gene is expressed under the control of the Aspergillus
nidulans trpC promoter and thus can be used as a dominant drug-resistance marker for the DNA-mediated
transformation of filamentous fungi. The successful application of both vectors was demonstrated by transforming
the homothallic ascomycete Sordaria macrospora as well as the β-lactam producer Acremonium chrysogenum. For
both fungi and for both vectors, transformation frequencies were between 10 and 40 transformants per 10 µg of
plasmid DNA.
Since the first successful DNA-mediated transformation of filamentous fungi (Case et al. 1979, Stahl et al. 1982)
several marker genes have been developed for selection of transgenic fungal strains (Fincham 1989, Lemke and Peng
1995). This includes the hph gene encoding the hygromycin B phosphotransferase which is applicable in
transformations without the need for constructing appropriate recipient strains. In order to generate an alternative
dominant marker gene that does not exhibit cross-resistance to hygromycin B, we have chosen the nat1 gene
conferring resistance to nourseothricin. The nat1 gene product is the nourseothricin acetyltransferase from
Streptomyces noursei (Krügel et al. 1993) which has been successfully used in the transformation of several yeast
species including Saccharomyces cerevisiae, Schizosaccharomyces pombe, Candida albicans and Cryptococcus
neoformans (Goldstein and McCusker 1999, McDade and Cox 2001, Hentges et al. 2005, Shen et al. 2005).
Here, we present the construction of two transformation vectors, carrying the commercially available nat1 gene
(Werner BioAgents, Jena, Germany) that are suitable for the transformation of filamentous fungi. As a first step in
establishing the nat1-based transformation system, sensitivity against the antibiotic nourseothricin (Werner
BioAgents, Jena, Germany) was tested for A. chrysogenum and S. macrospora. For A. chrysogenum and S.
macrospora, growth was inhibited on solid media at nourseothricin concentrations of 25 µg and 50 µg/ml,
respectively. In the following, the construction of two transformation vectors, which can be used for different
experimental purposes, is described.
First, the bacterial nat1 gene was fused to a fungal promoter that allows the expression of the nat1 gene in a wide
range of filamentous fungi. The nat1 gene was amplified with primer pair nat1-1 (5`-AACCATGGCCACCCTCGACGACACGGC-3`) and nat1-2 (5`-TAGCGGCCG
CTCAGGGGCAGGGCATGCTCATG-3`) using plasmid pHN15 (Werner BioAgents, Jena, Germany) as template.
The resulting amplicon is flanked by single NcoI and NotI restriction sites which were used for insertion into the
corresponding sites of vector pHAN (Kück, unpublished). pHAN is an expression vector that is suitable for use in
filamentous fungi. This vector carries a cloning site with NcoI and NotI recognition sequences that are flanked 5` and
3` by the A. nidulans trpC promoter and terminator, respectively (Mullaney et al. 1985). The resulting plasmid was
named pNAT2 (Kück and Godehardt, unpublished) and used as a source for the trpC promoter-nat1 fusion. The
following construction describes two vectors carrying this gene fusion. We found out that the trpC terminator
sequence is not necessary for efficient expression of the nat1 gene in the fungal hosts.
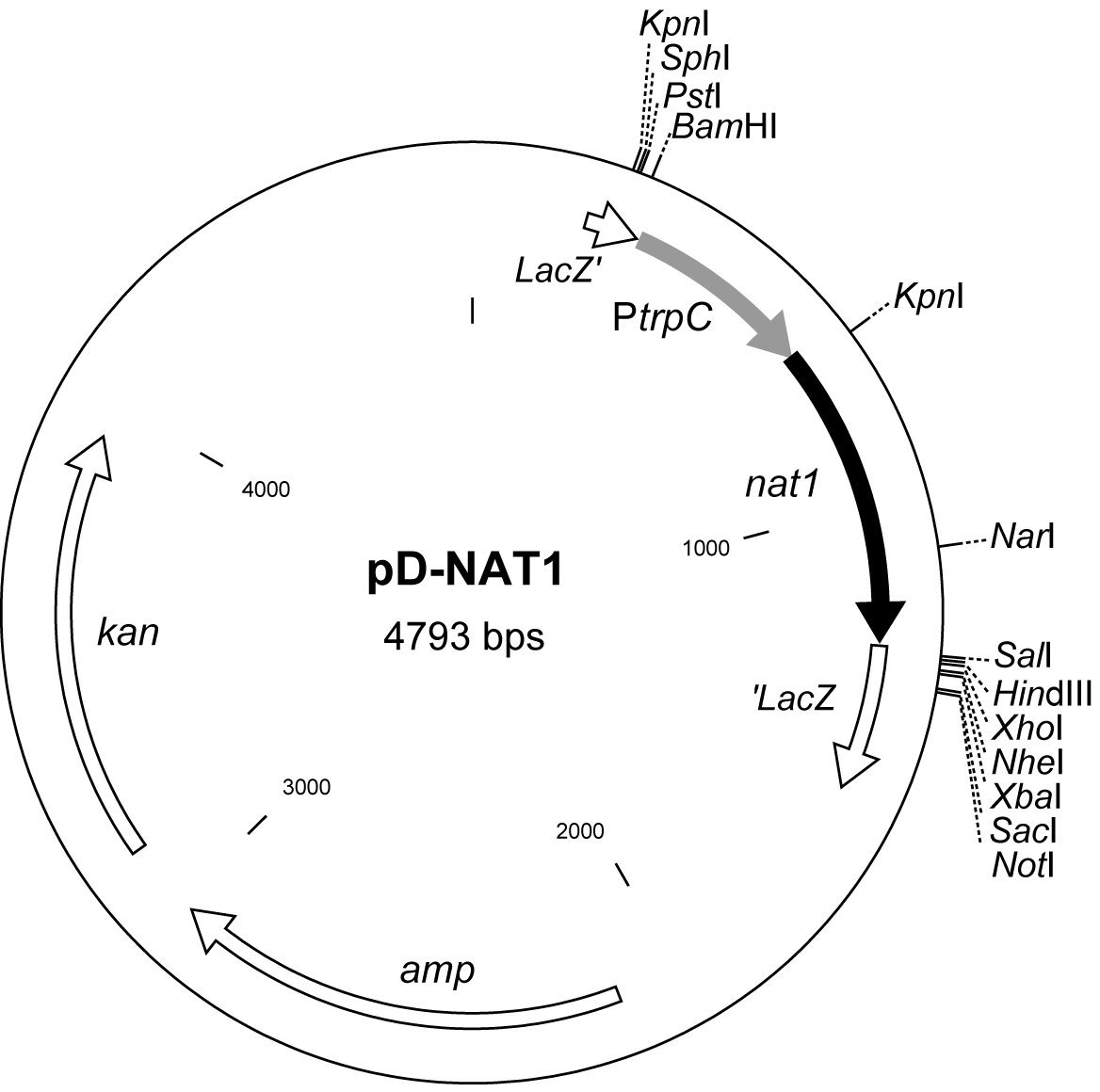
Vector pD-NAT1 (Fig. 1) was constructed using primers nat1-3 (5`-GAATTCAACTGA TATTGAAGGAGCA-3`)
and nat1-4 (5`-GGGCCCTCAGGGGCAGGGCATGCTCATGT AGA-3`) and the pNAT2 plasmid to generate the
PtrpC-nat1 amplicon which was inserted into cloning vector pDrive (Qiagen, Hilden, Germany). In this vector, the
nat1 gene is flanked on both sites by multiple cloning sites that can be used for directed insertion of fungal genomic
sequences to construct gene disruption strains by homologous recombination. Using previously reported
transformation procedures for A. chrysogenum and S. macrospora, the transformants were kept without selection for
24 hours (Kück et al. 1989, Walz and Kück 1995). The germinated protoplasts were subsequently overlaid with top
agar containing nourseothricin concentrations of 25 µg/ml (Ac) and 50 µg/ml (Sm). After 7 (Ac) or 2 (Sm) days,
transformants were transferred on solid rich medium with the above mentioned nourseothricin concentrations. We
obtained with vector pD-NAT1 frequencies of about 20 to 40 transformants per 10 µg of DNA, which is comparable
to the rather low frequencies that are obtained with the hph gene (Kück et al. 1989, Walz and Kück 1995). Most
importantly, the transformants did not show cross-resistance neither to hygromycin B nor to phleomycin.
|
|
|
Figure 1. Physical map of vector pD-NAT1. Kan and
amp refer to the kanamycin and ampicillin resistance
markers which can be used for selection of E. coli
transformants. The nat1 gene is fused with the fungal
trpC promoter from A. nidulans (Mullaney et al. 1985)
|
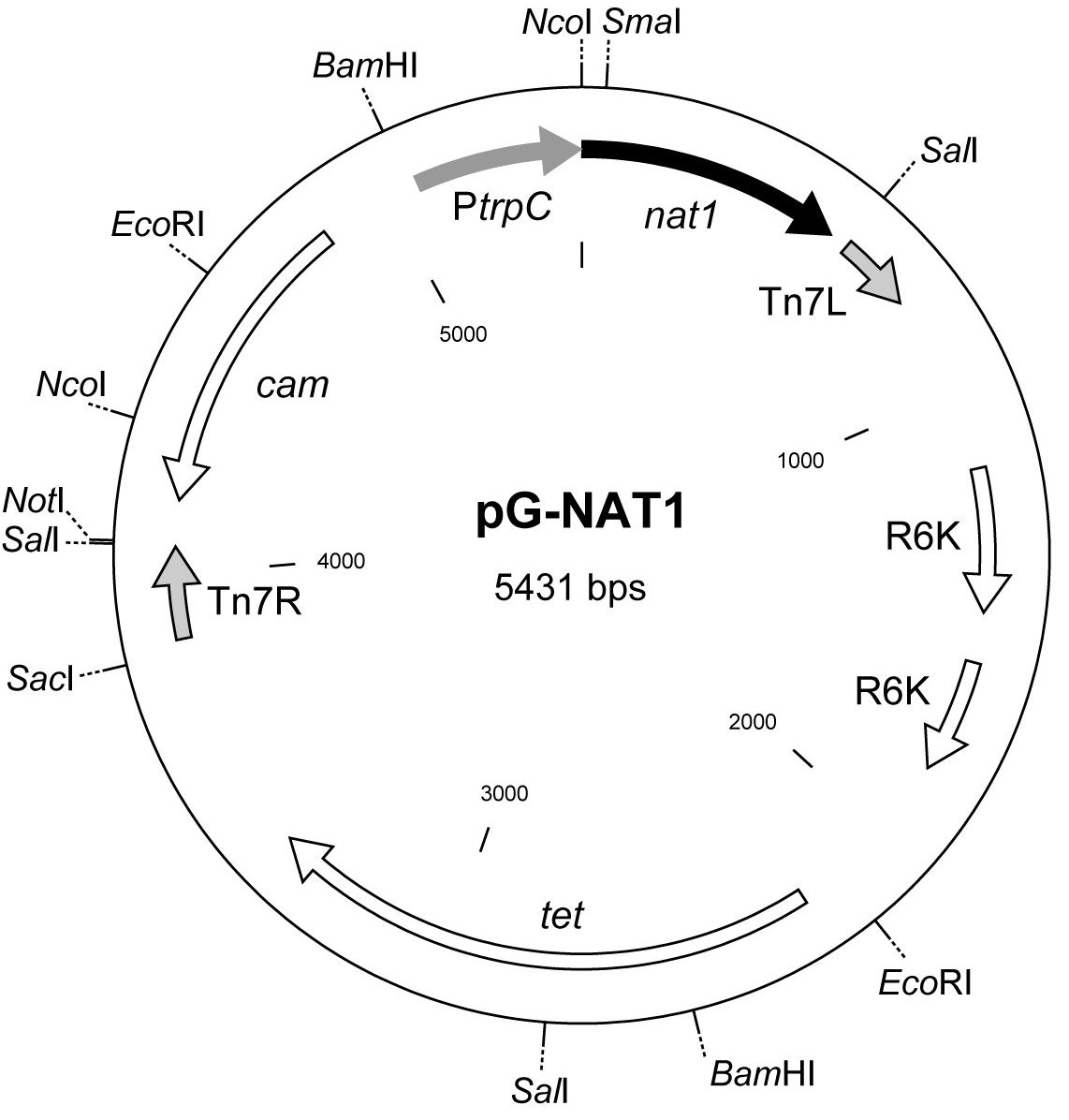
Figure 2. Physical map of vector pG-NAT1. Tet and
cam refer to the tetracycline and chloramphenicol
resistance markers which can be used for selection of
E. coli transformants. The nat1 gene is fused with the
fungal trpC promoter from A. nidulans (Mullaney et al.
1985). Tn7R and Tn7L indicate the right and left
TransprimerTM of transposon Tn7 and R6K marks the
E. coli R6Kg origin of replication (New England
Biolabs, USA)
|
To allow the nat1 gene to be used in further applications, for example, disruption of genes that can be used to
generate fungal knock-in strains, plasmid pG-NAT1 (Fig. 2) was generated. This vector carries a modified
TransprimerTM element derived from pGPS2.1. This plasmid is part of the GPSTM-1 Genome Priming System (New
England Biolabs, USA) which provides a simple in vitro method for generating a population of recombinant
plasmids (or cosmids) with randomly inserted TransprimerTM elements. The resulting disrupted genes are flanked by
large border sequences that promote homologous recombination in fungal recipient strains. For construction of pG-NAT1, a 1.3 kb EcoRI-NotI fragment from pNAT2 was treated with Klenow polymerase to generate blunt ends. This
fragment was inserted into the unique SwaI site within the Transprimer-2TM element of plasmid pGPS2.1 resulting in
the generation of pG-NAT1. In addition to the nat1 gene for selection in filamentous fungi,
pG-NAT1 also carries the cam and tet genes as selectable markers for E. coli transformation. Successful application
of this plasmid was shown when the Transprimer-2TM element of pG-NAT1, carrying the nat1 gene, was randomly
inserted into cosmid
clones carrying about 40 kb of fungal genomic sequences (Dreyer, Engh and Kück, unpublished data). The resulting
plasmids, isolated from E. coli, had a size of about 50 kb and were successfully transformed into A. chrysogenum
and S. macrospora at a frequency of about 20 transformants per 10 µg DNA. A similar plasmid to pG-NAT1 was
recently described by Jadoun et al. (2004) and instead of the nat1 gene, it carries the pyr4 gene, which can be used
for transformation of the appropriate uracil auxotrophic recipient strains. In the case of pG-NAT1, any fungal
recipient strain showing sensitivity against nourseothricin can be used for DNA-mediated transformation.
In conclusion, the two vectors described here will provide useful alternatives when other than the commonly used
resistance marker genes will be required for DNA-mediated transformation of filamentous fungi. For example,
multiple tagged or deleted strains can be constructed and in the case when simultaneous imaging of multiple proteins
is desirable, several reporter genes can be introduced.
Acknowledgements
We thank Ingeborg Godehardt, Kerstin Kalkreuter, Stefanie Mertens and Susanne Schlewinski for skilful technical
assistance, Danielle Janus and Malin Jansson for testing nourseothricin sensitivity, and Jacqueline Dreyer, Ines Engh
and Stefanie Pöggeler for discussion. Funding for this research was provided by the Deutsche
Forschungsgemeinschaft (SFB480-A1) and Sandoz GmbH (Kundl, Austria).
References
Case, M.E., M. Schweizer, S.R. Kushner, and N.H. Giles. 1979. Efficient transformation of Neurospora crassa by
utilizing hybrid plasmid DNA. Proc. Natl. Acad. Sci. USA 76: 5259-5263.
Fincham, J.R. 1989. Transformation in fungi. Microbiol. Rev. 53: 148-170.
Goldstein, A.L., and J.H. McCusker. 1999. Three new dominant drug resistance cassettes for gene disruption in
Saccharomyces cerevisiae. Yeast 15: 1541-1553.
Hentges, P., B. Van Driessche, L. Tafforeau, J. Vandenhaute, and A.M. Carr. 2005. Three novel antibiotic marker
cassettes for gene disruption and marker switching in Schizosaccharomyces pombe. Yeast 22: 1013-1019.
Jadoun, J., Y. Shadkchan, and N. Osherov. 2004. Disruption of the Aspergillus fumigatus argB gene using a novel in
vitro transposon-based mutagenesis approach. Curr. Genet. 45: 235-241.
Krügel, H., G. Fiedler, C. Smith, and S. Baumberg. 1993. Sequence and transcriptional analysis of the nourseothricin
acetyltransferase-encoding gene nat1 from Streptomyces noursei. Gene 127: 127-131.
Kück, U., M. Walz, G. Mohr, and M. Mracek. 1989. The 5´-sequence of isopenicillin N-synthetase gene (pcbC)
from Cephalosporium acremonium directs the expression of the prokaryotic hygromycin B phosphotransferase gene
(hph) in Aspergillus niger. Appl. Microbiol. Biotechnol. 31: 358-365.
Lemke, P.A., and M. Peng. 1995. Genetic manipulation of fungi by DNA-mediated transformation. In: Kück U (eds.)
The Mycota II Genetics and Biotechnology. Springer-Verlag, Berlin, Heidelberg pp. 109-139.
McDade, H.C., and G.M. Cox. 2001. A new dominant selectable marker for use in Cryptococcus neoformans. Med.
Mycol. 39: 151-154.
Mullaney, E.J., J.E. Hamer, K.A. Roberti, M.M. Yelton, and W.E. Timberlake. 1985. Primary structure of the trpC
gene from Aspergillus nidulans. Mol. Gen. Genet. 199: 37-45.
Shen, J., W. Guo, and J.R. Kohler. 2005. CaNAT1, a heterologous dominant selectable marker for transformation of
Candida albicans and other pathogenic Candida species. Infect. Immun. 73: 1239-1242.
Stahl, U., P. Tudzynski, U. Kück, and K. Esser. 1982. Replication and expression of a bacterial-mitochondrial hybrid
plasmid in the fungus Podospora anserina. Proc. Natl. Acad. Sci. USA 79: 3641-3645.
Walz, M., and U. Kück. 1995. Transformation of Sordaria macrospora to hygromycin B resistance: characterization
of transformants by electrophoretic karyotyping and tetrad analysis. Curr. Genet. 29: 88-95.
Return to the FGN 53 Table of Contents
Return to the FGSC main page